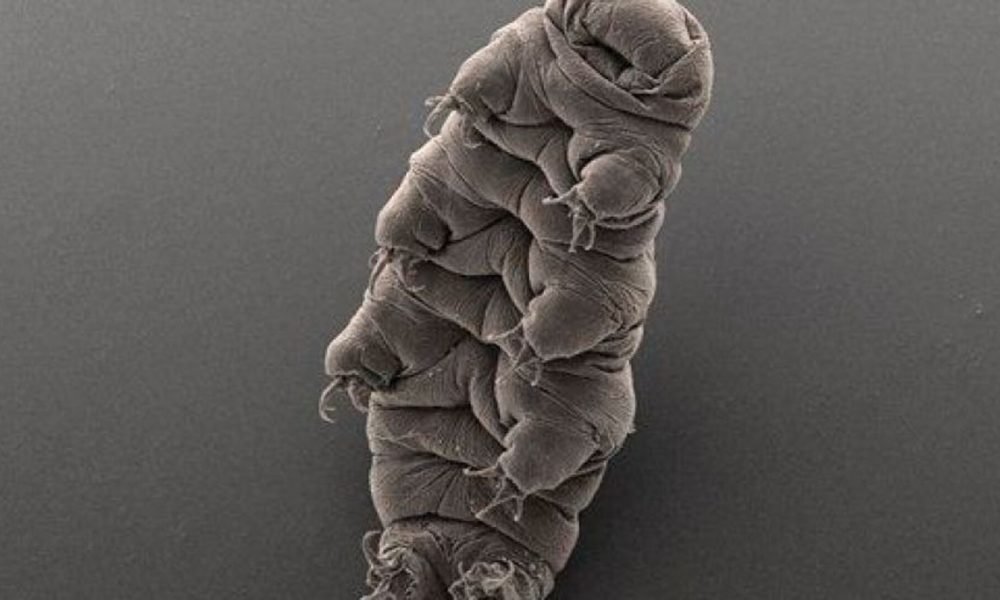
A tardigrade. (AP Photo/BobGoldstein && amp; Vicki Madden, UNC Chapel Hill)
Scientists just recently understood a essential component in tardigrades’ toolbox of superpowers, unraveling how a distinct protein secures everybody’s preferred tiny water bears from damaging radiation.
Though small, tardigrades are infamously hard. They can weather severe conditions that would eliminate most kinds of life, consisting of direct exposure to freezing cold, broiling heat, and the vacuum and deadly radiation of area.
But what are the chemical tricks that provide tardigrades their near-invulnerability?To response that concern, scientists peered carefully at a substance discovered just in tardigrades: the so-called damage-suppressor protein, or Dsup.
This protein’s protective powers were formerly discovered to extend beyond tardigrades; when contributed to human cells, Dsup safeguards versus damage from X-rays. And now, researchers have actually found how Dsup binds to chromosome structures and secures DNA from the damaging impacts of radiation, the scientists reported in a brand-new research study.
Related: 8 Reasons Why We Love Tardigrades
“We thought this fascinating protein in an extreme organism might tell us something new that we wouldn’t get from regular proteins,” stated research study co-author James Kadonaga, a teacher with the Division of Biological Sciences at the University of California, San Diego.
Though tardigrades might appear unbreakable, they need water in order to be active and replicate. In water’s lack, they pull back into a type of suspended animation called a tun state, expelling wetness from their bodies and existing in a desiccated limbo up until more-hospitable conditions return.
As tuns, tardigrades are resistant to a lot of kinds of damage and can even be restored after years, potentially even after hanging around on the moon. Thousands of tuns might have been spread on the lunar surface area after the Israeli lunar lander Beresheet (which was bring a payload of desiccated water bears) crashed on April 11 throughout a stopped working landing effort. Under particular conditions, if they made it through the crash landing, those freeze-dried tardigrades could still return to life, LiveScience formerly reported.
Seemingly unbreakable
Some of the proteins that enable tardigrades to restore after being dried are discovered in other organisms, however Dsup is special to water bears. And while previous research studies discovered that this protein made human cells resistant to X-ray radiation, the systems of how Dsup did that doubted.
In the brand-new research study, the scientists found that Dsup binds to a structure called chromatin, a bundle that holds a cell’s long hairs of DNA in a thick bundle, Kadonaga informed Live Science.
“We found it binds to chromatin. Then we asked, ‘How does it make it resistant to X-rays?'” he stated.
When cells are bathed in X-rays, water particles split and type extremely reactive particles of oxygen and hydrogen called hydroxyl radicals; these radicals can harm DNA inside cells, according to the research study.
“We thought, ‘Why don’t we just see if Dsup can protect DNA from hydroxyl radicals?’ And the answer is yes, it can,”Kadonaga described. High- energy Dsup has a cloud-like structure; the cloud surrounds the DNA’s chromatin envelope, obstructing hydroxyl radicals and avoiding them from interfering with cellular DNA, the scientists reported.
“Now that we know how it worked, that’s a stepping stone to potentially using it for practical applications,”Kadonaga stated.
By piecing together how Dsup operates at ever-more-precise levels, researchers can then utilize it as a plan for constructing other kinds of proteins– “better versions of Dsup”– that are a lot more reliable at safeguarding cells from DNA damage, Kadonaga stated. These brand-new proteins most likely will not be utilized to produce radiation-proof individuals, however they could enhance the strength of cultured cells that are utilized for growing pharmaceuticals, he included.
“You can have more-durable cells, more-longer-lived cells. That might be a case for putting some form of Dsup in that cell,” he stated.
The findings were released online Tuesday (Oct 1) in the journal eLife.
Originally released on LiveScience.